Introduction
The complement system is part of the immune defense with a number of biological functions, many of which contribute to the inflammatory reaction by activation of peripheral blood mononuclear cells (PBMCs) and endothelial cells. An intact complement system is required for protection against infection and for maintaining internal tissue homeostasis.
However, the system is a “double-edged sword” because improper, enhanced, or uncontrolled activation is disadvantageous and potentially harmful for the patient. Complement comprises more than 30 proteins acting together in a highly specific manner and is kept under strict control by regulatory proteins.(1) The system can be activated by three initial pathways described below:
| Classical | Activated when natural or elicited antibodies bind to antigen, but it can also by activated independent of antibodies by a number of substances, like C-reactive protein (CRP), when bound to the surface of a cell |
| Lectin | initiated by mannosebinding lectin (MBL), ficolins 1–3, and several collectins, all of which recognize conserved structural patterns on microbes and on various structures on damaged self cells. MBL is homologous to C1q and triggers three MBL-associated serine proteases (MASPs), of which MASP-1 and MASP-2 contribute in further downstream complement activation.(2) Subsequent lectin pathway steps are virtually identical to classical pathway activation by forming the same C3 and C5 convertases |
| Alternative | Activated by damaged cells or by foreign cells and is facilitated by the continuous spontaneous hydrolysis of C3. The alternative pathway also has an important function in the complement system by providing an amplification loop that enhances C3 activation independently of which pathway is initially activated. This effect is mainly due to properdin (FP), the only positive regulator in the complement system, which stabilizes the C3 convertase |
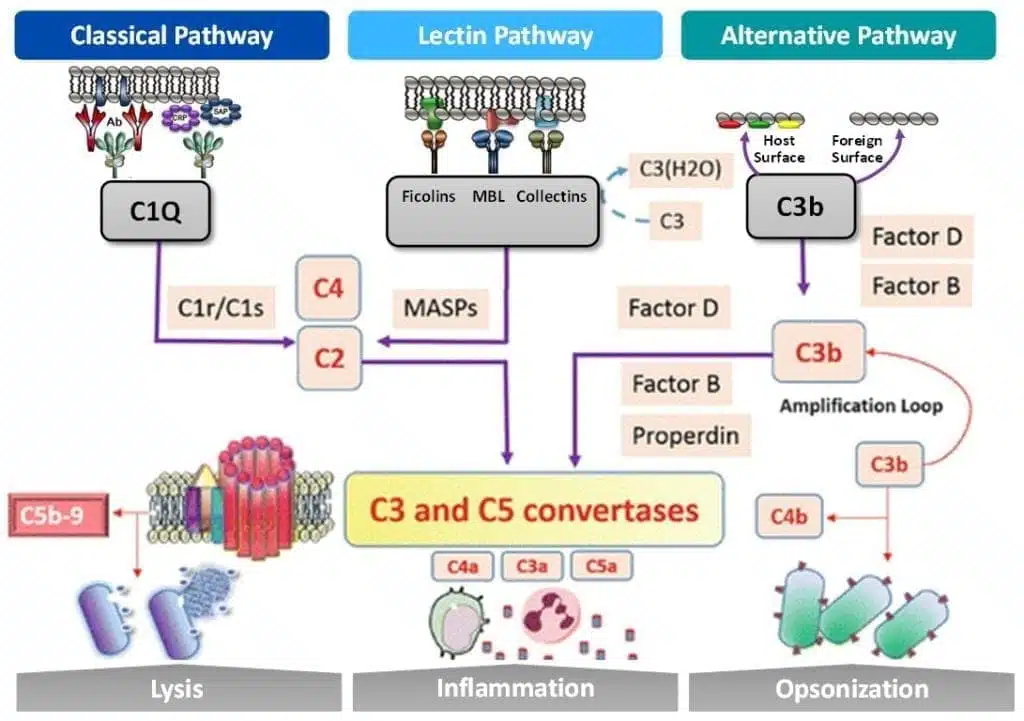
The complement system can be activated through three pathways indicated above. All pathways converge on C3 and C5 convertases leading to downstream lysis, inflammation and opsonization.
Key Functions and Mechanisms
The complement system is highly potent and intended to act locally to protect the host against danger. External dangers like pathogenic microbes express pathogen associated molecular patterns (PAMPs) that are recognized by the host’s pattern recognition molecules/receptors, including PBMCs. The initial complement components are typically PBMCs that react with the pathogen and initiate the subsequent response to fight infection. Complement deficiencies are thus associated with increased risk of infection.
The mechanisms of the complement system contribute to an inflammatory reaction in the tissue, which is a hallmark of complement activation. It is, however, of utmost important for the host to restrict the activation of the complement system, and the defense mechanisms should act locally. As soon as an infection or other activation of the complement cascade spreads and induces a systemic activation, the host will be more threatened by self-destructive uncontrolled complement activation than by the microbe itself. This is similar to the avoidance of uncontrolled activation of the coagulation system to avoid disseminated intravascular coagulation.
In addition to recognizing exogenous PAMPs to fight infection, the complement immune cells also sense endogenous danger from damage-associated molecular patterns (DAMPs) that are exposed upon tissue damage from events such as ischemia reperfusion (e.g., myocardial infarction, stroke, and transplantation), trauma, and other sterile inflammatory conditions. Molecules that are normally hidden and molecules that change their structure (neoepitopes), become exposed to the complement components upon tissue damage. Such structures can be recognized by naturally occurring IgM antibodies that activate the classical pathway, by the lectin pathway PRM, or by the alternative pathway when discriminating an intact self-surface from a damaged or foreign surface.(2) The sterile inflammation occurring due to damaged self-tissue might induce tissue damage and organ dysfunction, and in case of systemic activation, such as multiple trauma, a systemic inflammatory response might occur leading to multi-organ failure and death.
The complement system is thus dichotomous, serving as both defender against pathogens and the primary cause of life-threatening inflammation depending on the actual circumstances. Most importantly, it does not play alone, but is part of a complex array with a number of other biological systems, including the other plasma cascades and the other branches of innate immunity, including toll-like receptors, as well as with metabolic and neuroendocrine systems.
Diseases from Dysregulation of the Complement System
A role for complement in the pathogenesis of several neurodegenerative diseases has been suggested, including Guillain-Barre syndrome and neurodegenerative disorders such as Alzheimer’s disease, multiple sclerosis, and amyotrophic lateral sclerosis. Two diseases of particular interest are associated with autoantibodies, and activation occurs through the classical pathway. The first is myasthenia gravis in which patients present with autoantibodies to the acetylcholine receptor, which impair neuromuscular transmission signals. The second is neuromyelitis optica, which was previously regarded as a subgroup of multiple sclerosis with vision impairment but is now known to be a separate entity where the pathogenic factor is autoantibodies against aquaporin 4. These two diseases might benefit from complement inhibition, e.g. with a blocking antibody to C1s to halt activation at an early step, or with a C5 inhibitor because C5b-9 is probably crucial for the tissue damage that occurs in this disease. Clinical trials of complement inhibition are ongoing in myasthenia gravis patients.
Age-related macular degeneration (AMD) is the most common cause of blindness in industrialized countries and is closely related a dysregulation of the alternative pathway from genetic mutations. Clinical trials for blocking factor D are underway. There is also evidence for a role of the complement system in chorioretinitis and possibly also in glaucoma.
These diseases are just a sample of some of the conditions for which human or animal studies support a role for the complement system and thus might be candidates for future therapy. However, the list is much longer than this, including diseases like ANCA vasculitis, catastrophic antiphospholipid syndrome, cold-agglutination, lung diseases, allergies, atherosclerosis, ischemia-reperfusion injury like myocardial infarction and stroke, trauma, sepsis leading to systemic inflammatory response syndrome, and many more.
Therapeutics Related to the Complement System
The only drug targeting the complement system in clinical use today is eculizumab, which inhibits the activation of C5. Other complement inhibitors are in clinical trials, and potential new drugs are continuously being developed. Specific inhibition of the initial pathways might be obtained, for example, by targeting C1s (the classical pathway), MASP-2 (the lectin pathway), or factor D or factor B (the alternative pathway). C3 is a potent component to target because it is the first common molecule for all three initial activation pathways. C3aR might also be a possible target in certain conditions. Other C5 inhibitors are also in clinical trials, including agents that block C5 cleavage, like eculizumab, or block C5a or its receptors C5aR1/2, with the latter approaches leaving the C5b-9 pathway open to form the complex.(3) Complement tests will be increasingly important because complement inhibition is expanding in the clinic and novel inhibitors are being developed. Many tests are already available as described above, and these will be useful for testing the efficacy of potential inhibitors both in vitro and in vivo.
The functional activity measurement of C5b-9 deposition will be particularly useful because it covers the whole complement cascade. Thus, blocking the function of any of the ordinary native components can be evaluated using this test, with the added advantage of the test being standardized, rapid, and easy to perform. Inhibitors blocking complement receptors (e.g., C5aR1) and/or activation products (e.g., C5a) will, however, require the development of novel assays for testing their efficacy. Future pharmacological treatment will in general focus on individualized therapy, such as giving optimal doses to obtain effective treatment, avoiding adverse effects, and reducing cost.
Cytologics supports biotech and pharmaceutical customers developing therapies related to the complement system. We supply human PBMCs and purified immune cells that are critical for drug discovery and preclinical research. We also work closely with diagnostic companies that design and manufacture complement assays for clinicians. If you have questions on how our products & services can advance your research, then contact us now to speak with one of our scientists.
References
(1) Dunkelberger, J., Song, WC. Complement and its role in innate and adaptive immune responses. Cell Res 20, 34–50 (2010). https://doi.org/10.1038/cr.2009.139
(2) Janeway CA Jr, Travers P, Walport M, et al. New York: Garland Science; 2001.
(3) Ricklin D, Lambris JD. Complement-targeted therapeutics. Nat Biotechnol. 2007;25(11):1265-1275. doi:10.1038/nbt1342.